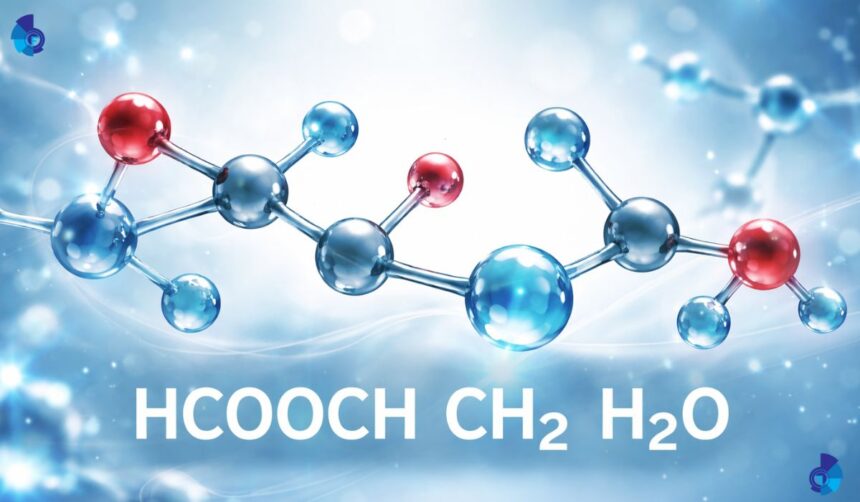
Chemical formulas can look complex, but most of them tell a story. The expression HCOOCH CH2 H2O represents a chemical arrangement involving a formate group, a carbon chain component, and water. When these pieces come together, they illustrate the way molecules can connect or separate depending on temperature, environment, or reaction type. The purpose of this article is to unpack these ideas in a clear and helpful way. Instead of focusing on narrow technical explanations, the discussion will highlight real-world relevance, simple explanations of composition, and the broader value of studying reactions built around this molecular structure.
In many discussions, the combination is used to illustrate how a molecule can break down or take on different shapes during processes like hydration, decomposition, or heating. This makes the compound useful as a teaching tool and a practical reference for industries that work with organic materials. Understanding its composition also helps readers see how different chemical elements influence stability, function, and behavior in everyday materials.
Understanding the Composition of HCOOCH CH2 H2O
At its core, the expression HCOOCH CH2 H2O brings together three recognizable components. The first is a formyl or formate-related structure, which is common in different organic molecules. The second is a carbon chain portion represented by CH2, which appears in countless organic reactions. The third is water, a molecule that plays a vital role in shaping how many compounds behave. When examined together, the formula serves as a simplified representation of a hydrated or partially bonded material, depending on the environment in which it appears.
In a general sense, the composition illustrates the relationship between organic groups and water. Hydration can stabilize molecules in some settings or cause them to break apart in others. Because the structure includes a water molecule, it also points to potential interactions where water participates actively instead of merely surrounding the substance. This makes the formula helpful for understanding how certain organic groups react under various conditions such as heating, mixing, or exposure to catalysts.
How Environmental Conditions Influence the Structure
Chemical behavior is rarely fixed. The combination represented by HCOOCH CH2 H2O may appear stable under certain conditions, but changes in heat, pressure, or acidity can influence its form. For example, heating can break internal bonds and create simpler molecules, while cooling can allow the components to remain loosely associated. Water also plays a major role in determining how the compound behaves because it interacts through hydrogen bonding and influences stability.
When exposed to acidic or basic conditions, the structure may shift even more. In acidic environments, the molecule may undergo reactions that remove or rearrange the water portion. In basic environments, the molecule may break apart more easily, releasing smaller components into the mixture. These behaviors make the formula important for industrial research, educational demonstrations, and laboratory testing.
Reactions Involving HCOOCH CH2 H2O
Reactions involving this compound often focus on hydration, decomposition, and transformation. Hydration reactions refer to processes where water takes part in rearranging or stabilizing molecular structures. Because the formula includes water, it can help illustrate how water becomes part of a reaction rather than acting as a simple solvent.
Decomposition reactions may occur when the compound is heated. In these reactions, the molecule may break down into simpler forms such as carbon dioxide, smaller organic fragments, or other gases depending on the temperature and environment. These transformations show how molecular structures are not static and can evolve as conditions change.
Another type of reaction involves substitution or exchange, where one part of the molecule is replaced or modified by another substance. This kind of reaction helps industries create new chemicals by modifying existing structures. The presence of water makes certain reactions more likely because water can assist in breaking existing bonds and forming new ones through hydrolysis.
Why Hydration Matters in Organic Chemistry
Hydration is one of the most important concepts in organic chemistry, and the expression HCOOCH CH2 H2O helps demonstrate why. Water can influence structure, stability, and reaction speed. When a molecule takes on water, it sometimes becomes more stable and easier to work with. In other cases, hydration can trigger decomposition or transformation, revealing new stages in the life cycle of a compound.
In natural environments, hydration is a major factor in how materials break down. Many compounds that appear stable in dry conditions become reactive when water is introduced. This is one reason why industries pay close attention to moisture levels when storing or transporting chemicals. The formula highlights how water becomes part of the internal structure and cannot be ignored when studying chemical behavior.
Industrial Importance of Molecules with Similar Structures
Although the exact expression represents a conceptual or simplified form, molecules with similar compositions are common in manufacturing, agriculture, materials science, and energy-related processes. Compounds containing formate groups, carbon chains, and associated water molecules play important roles in creating fuels, coatings, adhesives, and biodegradable materials.
In manufacturing, similar structures appear during the production of solvents and intermediates used to make plastics, textiles, and resins. Their predictable behavior helps engineers design processes that remain stable across different temperatures and storage conditions. In agriculture, related compounds appear in fertilizers and treatments that release nutrients or protective components gradually when exposed to moisture.
Benefits of Studying the Chemical Behavior of This Compound
One of the clearest benefits of studying HCOOCH CH2 H2O is gaining insight into how organic molecules behave when interacting with water. Water’s involvement in chemical reactions is sometimes overlooked because it appears so common, but it plays a major role in controlling speed, direction, and outcome. By studying compounds that include water in their structure, researchers can design materials that respond more predictably to environmental conditions.
Understanding these reactions also allows researchers to improve safety. Many accidents in laboratories or industrial facilities occur when materials react unexpectedly with moisture. Studying formulas that illustrate water’s effect on organic compounds helps reduce risk by showing how hydration can influence stability.
The study of such compounds also supports innovation. When scientists know how water affects organic materials, they can design new compounds for packaging, medicine, and energy applications. These new materials can be stronger, safer, or more efficient because their creators understand how hydration influences chemical structure.
Role in Educational and Theoretical Chemistry
The formula is also valuable in teaching environments. Students often struggle to understand how water can both help and disrupt organic structures. Using a formula like HCOOCH CH2 H2O enables instructors to explain how hydration changes molecular shape, energy levels, and reaction pathways. This helps students build a practical understanding of organic chemistry rather than seeing it as a series of disconnected facts.
In theoretical chemistry, the formula supports discussions about reaction mechanisms. It helps illustrate how molecules move, where bonds break, and why certain outcomes occur. Researchers can use it as a model structure when designing simulations or predicting reaction behavior under specific conditions.
How the Compound Helps Explain Reaction Pathways
Reaction pathways describe how a chemical transformation moves from start to finish. A compound that includes water in its structure can help explain multiple steps in these pathways. Water may enter early in the process, influence the middle stages, or help form the final products. Studying such structures helps chemists understand why certain products are favored over others.
This understanding is especially important in industries that rely on selective reactions. If the wrong product forms, the entire process may fail. Using compounds like this as teaching tools or reference points allows chemists to improve reaction design and troubleshoot unexpected results.
Potential Applications in Material Development
Some materials require controlled hydration to achieve the right texture, strength, or flexibility. Understanding how hydration changes internal structure allows manufacturers to create coatings, films, and binders that respond predictably to moisture. While the formula here is conceptual, it reflects the kind of behavior needed to design smart materials that absorb or release water in a controlled manner.
These materials may appear in construction, packaging, and environmental technology. For example, moisture-sensitive materials can help regulate humidity or protect products from damage during shipping. Each application benefits from understanding the relationship between water and organic molecular structures.
Environmental Relevance of Hydrated Organic Compounds
Organic compounds that interact with water also play important roles in natural ecosystems. Water exposure helps break down certain materials into smaller fragments that become nutrients or foundational pieces for new compounds. Studying hydration in molecules like HCOOCH CH2 H2O helps environmental researchers understand how natural degradation happens and how long different substances remain in soil, air, or water.
Understanding hydration also supports cleaner manufacturing. When industries recognize how water influences their materials, they can design processes that reduce waste, lower emissions, or improve biodegradability.
Final Thoughts
The expression HCOOCH CH2 H2O offers a clear window into the behavior of organic compounds when exposed to water. It illustrates how hydration affects stability, reaction pathways, and transformation. Studying such structures provides valuable insight for industries, educators, researchers, and anyone interested in how chemistry works behind the scenes. Whether used to explain theoretical concepts or improve practical applications, the compound helps demonstrate the essential role water plays in shaping the world of organic chemistry.
Frequently Asked Questions
1. What does HCOOCH CH2 H2O generally represent?
It represents a simplified way to show how organic molecules can include or interact with water. This combination helps illustrate hydration and the role water plays in altering chemical structures.
2. Why is water included in the formula?
Water appears because it participates directly in reactions involving hydration, stabilization, or transformation. It is not just a surrounding solvent but an active part of the structure.
3. Does this compound appear in real-world materials?
Compounds with similar structures are common in industrial materials, organic intermediates, and substances used in agriculture or manufacturing. They help show how organic groups behave when hydrated.
4. How does temperature affect the compound?
Rising temperature can cause the molecule to break down, while lower temperature allows it to remain more stable. Heating often triggers decomposition reactions.
5. Is this compound safe to handle?
Safety depends on the exact form and environment, but understanding hydration behavior helps reduce risks. Many organic compounds become more reactive when exposed to moisture.
6. Why is this formula useful in teaching chemistry?
It provides an easy example of how water influences molecular structure, helping students visualize hydration and reaction pathways more clearly.
7. What industries benefit from studying compounds like this?
Manufacturing, materials science, environmental research, and agriculture all gain from understanding how water affects organic molecules and their stability.
For More: fogmagazine.co.uk